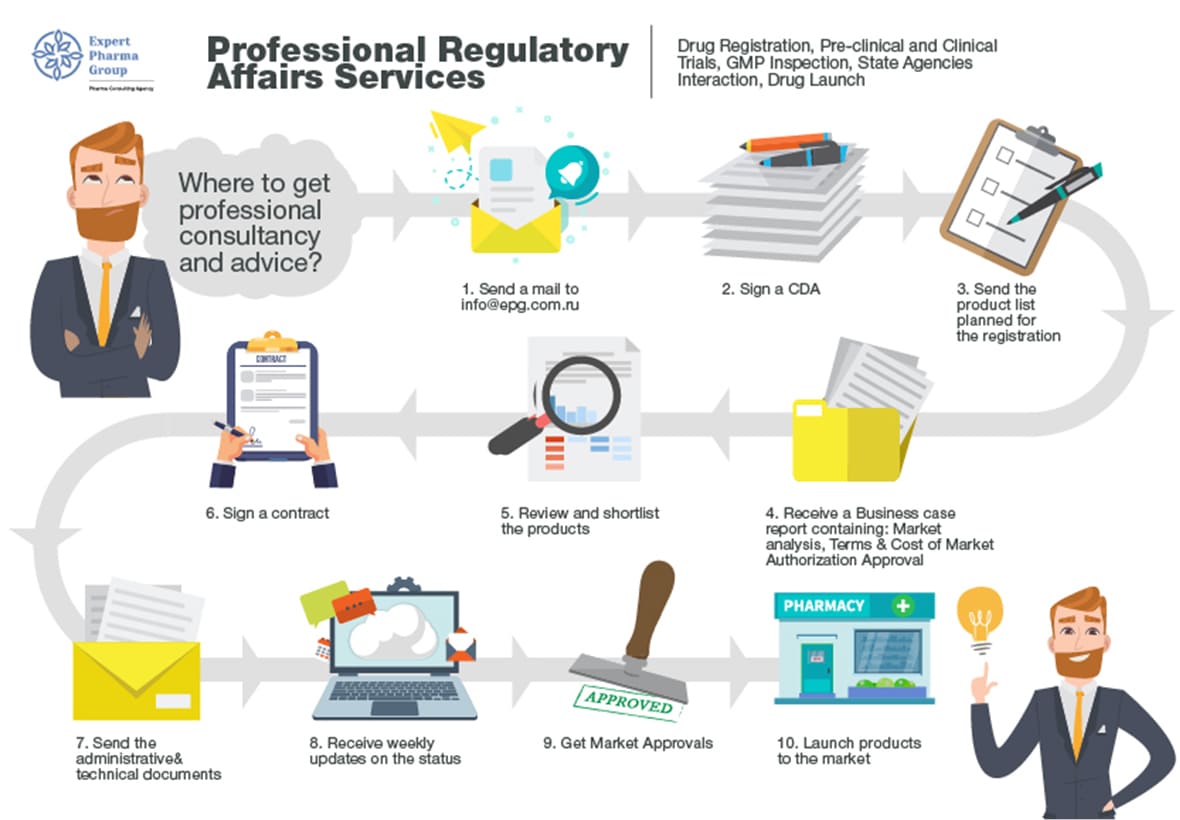
China CDE Adjusts Notarization Document Requirements For Overseas Drug Registration During Covid-19 - Accestra Consulting
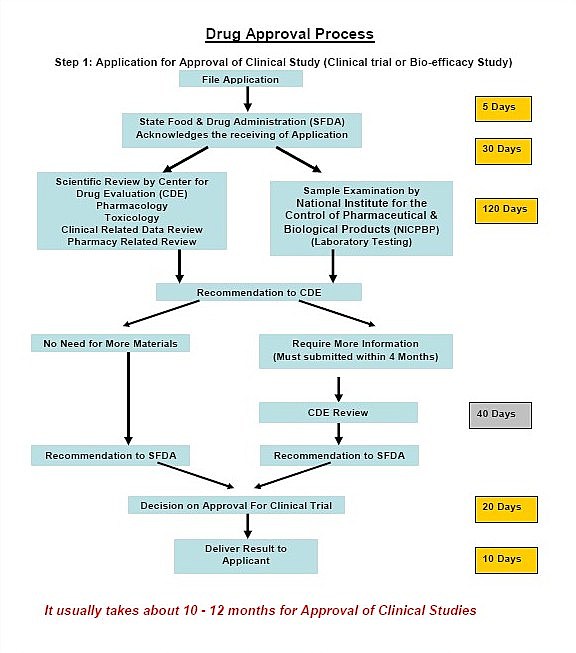
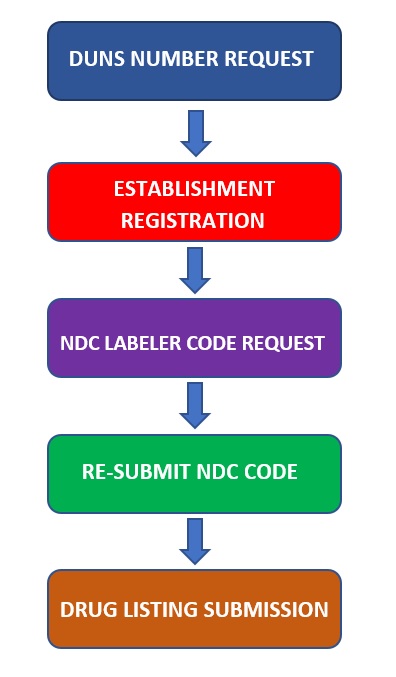
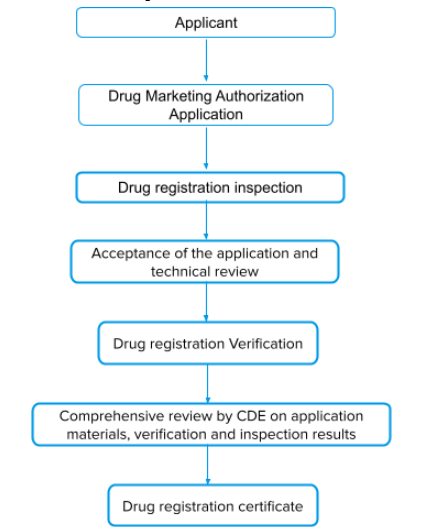
The procedure for Import Drug SFDA Registration-China FDA,SFDA,CFDA,MOH,MOA,AQSIQ,CNCA,CIQ registration approval license for cosmetics,health food supplement,medical device,IVD,drug,infant milk powder,dairy,pet food ,disinfectant etc.

The procedure for Import Drug SFDA Registration-China FDA,SFDA,CFDA,MOH,MOA,AQSIQ,CNCA,CIQ registration approval license for cosmetics,health food supplement,medical device,IVD,drug,infant milk powder,dairy,pet food ,disinfectant etc.










![Generic drug approval process in INDIA [12] | Download Scientific Diagram Generic drug approval process in INDIA [12] | Download Scientific Diagram](https://www.researchgate.net/publication/325145465/figure/fig2/AS:626451065872387@1526368981583/Generic-drug-approval-process-in-INDIA-12.png)
.png)